Share this post:
Author: Brent N. Reed, PharmD, BCCP, FAHA
Despite being the mainstay of therapy, questions remain as to how to properly use loop diuretics in patients with acute decompensated heart failure (ADHF). In this post, we’ll cover four of the most common mistakes with using loop diuretics in this population.
Mistake #1: Not giving a high enough dose.
Most people remember that the DOSE trial showed that high doses of intravenous (IV) loop diuretics (defined as 2.5-times patients’ home doses) improved congestion more rapidly than lower doses (1-times patients’ home doses) at the expense of a slight increase in the risk of transient worsening of renal function (WRF) (1). However, what most people don’t know is just how high the dose-increases in DOSE actually were. Since investigators used an arithmetical oral-to-IV conversion (i.e., not accounting for the approximately 50% bioavailability of furosemide), patients in the low-dose arm actually received twice their home dose (for example, 40 mg of oral furosemide transitioned to 40 mg IV is a two-fold dose increase). That means patients in the high-dose arm actually received five times their home dose. Despite such a considerable increase in dose, the only safety concern in the high-dose arm was transient WRF, which resolved by the end of the trial. These results corroborate other studies showing that transient WRF does not worsen long-term outcomes, suggesting that the perceived risks of high-dose diuretic therapy are often exaggerated (2).
A closely related mistake is increasing the frequency of dosing before increasing the dose. Since loop diuretics have a steep dose-response curve (see Figure 1), the threshold concentration for diuresis must be achieved before more frequent dosing will be helpful.
Bottom line: Don’t shy away from going “big” with loop diuretics upfront in ADHF, and find comfort in knowing that WRF is transient and amenable to dose decreases.
Figure 1. In order to elicit a diuretic response, the dose selected must exceed the diuretic threshold, regardless of how frequently it is given. Doses exceeding the ceiling dose do not improve diuresis.
Mistake #2: Not giving diuretics early enough (or giving vasodilators before loop diuretics).
Although congestion is by far the most common presentation of ADHF, it remained controversial for some time as to whether the most appropriate initial therapy should be diuretics or vasodilators. However, the vascular hypothesis has been subjected to multiple randomized controlled trials over the last several years, and early vasodilator use in ADHF has thus far failed to significantly improve outcomes (3–5). Additionally, given the exaggerated response to vasodilation in patients with heart failure with preserved ejection fraction (HFpEF), aggressive vasodilator use may actually compromise cardiac output and thus worsen outcomes in a subgroup that now comprises half of the population with ADHF (6, 7).
One of the most commonly cited arguments against early IV diuretic use in ADHF is the delay between drug administration and clinical response. While it’s true that diuresis does not occur until about 20-30 minutes after administration, what most people don’t realize is that high-dose loop diuretics actually improve congestion via functional venodilation within the first 5 minutes of administration (8). The resulting increase in venous capacitance enhances transcapillary refill rate, further contributing to the intravascular volume available for diuresis as concentrations in the nephron approach the diuretic threshold.
Bottom line: Most patients with ADHF present with congestion and excess fluid has to be eliminated somehow, so high-dose IV diuretics can both relieve acute symptoms in the short-term and eliminate volume in the long-term.
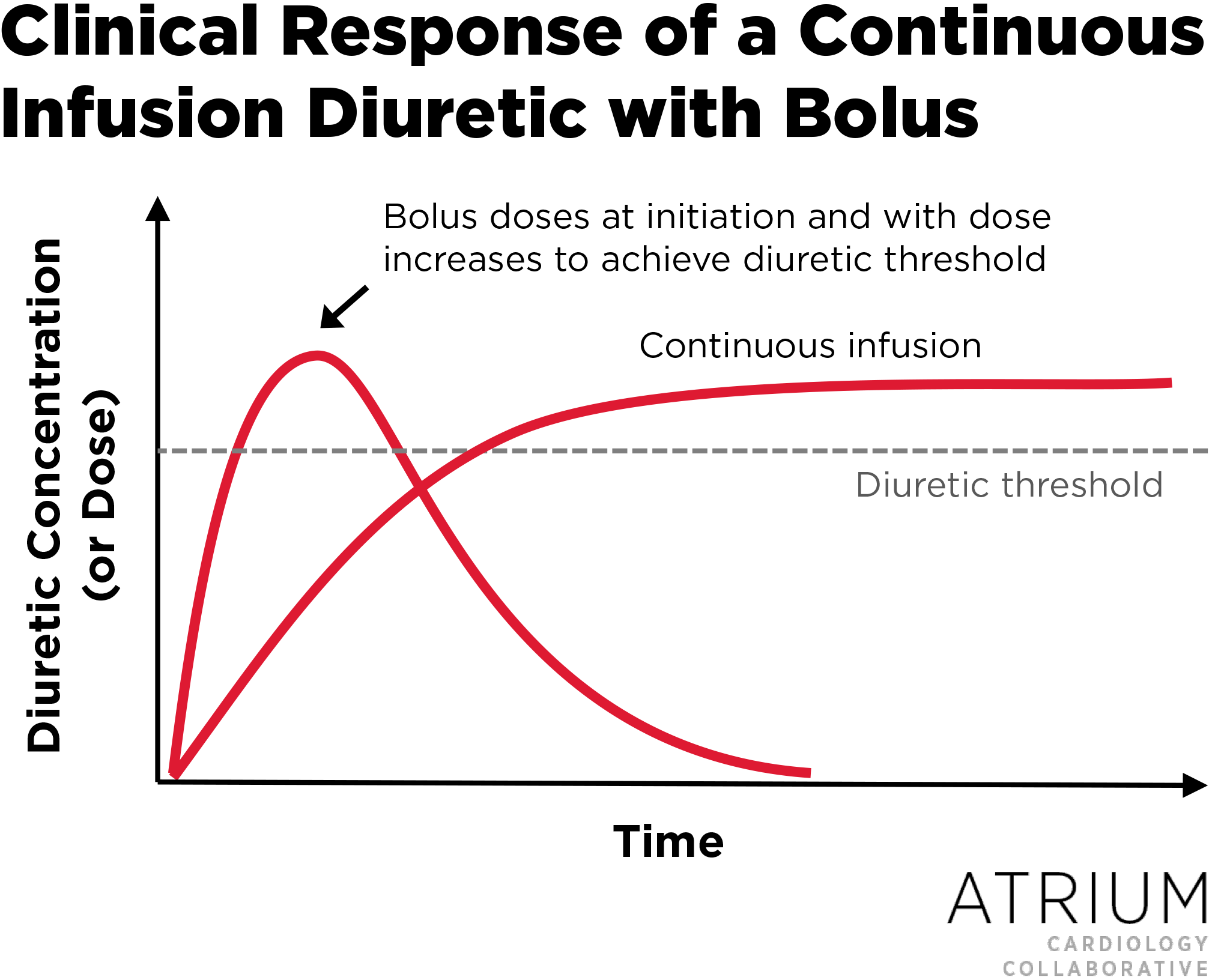
Mistake #3: Not giving a bolus when initiating or up-titrating a continuous infusion.
You may be wondering why we’re even discussing continuous infusions since the DOSE trial suggested they were no better at relieving congestion than IV boluses (1). While that might seem true on an initial review of DOSE, there is more to the story. As we discussed in a previous post, patients receiving therapy as IV boluses were twice as likely to have their doses increased or get a thiazide-type diuretic added to their regimen (p < 0.05 for both). However, it turns out that they not only got these extra boosts – they also got a head start as well. As was revealed in a recent review (9), patients in the continuous infusion arm of DOSE did not receive an initial bolus.
The reason this latter detail is important is that the full effect of a continuous infusion is not realized until it achieves steady state, when the concentration of drug administered equals the concentration eliminated (Figure 2). Given a half-life of 1-2 hours, steady state for a furosemide infusion doesn’t occur until 6-10 hours after the initiation of therapy. Fortunately, diuresis can be achieved earlier than steady state by administering a bolus at the initiation of an infusion and with any subsequent dose increases. Given a duration of action of approximately 6 hours, the bolus of furosemide should wear off at approximately the same time as the infusion achieves steady state. In terms of the bolus dose to administer, we typically select the amount that the patient will receive from the infusion over the next 6-10 hours (e.g., up to 100 mg if initiating an infusion at 10 mg/hour) up to a maximum of 200 mg.
Bottom line: To prevent a delay in response, administer a bolus when initiating or increasing the dose of a continuous infusion loop diuretic.
Figure 2. A continuous infusion diuretic must achieve steady state before a complete response is observed. Administering a bolus at the initiation of a continuous infusion (and with any dose increases) eliminates this gap in response.
Mistake #4: Not observing patients on their intended discharge regimen prior to discharging them.
Given the rising costs of care for hospitalized patients, teams are often pressured to discharge patients as soon as possible. As a result, a number of decisions are often made on the day of discharge, such as the re-initiation or titration of guideline-directed medical therapy. Another decision that is often deferred until the day of discharge is transitioning patients from IV to oral loop diuretic therapy. Although guidelines suggest that oral therapy be initiated prior to discharge in order to confirm efficacy and safety, no data existed to support this recommendation until just recently (7, 10, 11). In an observational study of patients with ADHF admitted to our cardiology services, we evaluated whether monitoring patients on their intended discharge diuretic for less than 24 hours was associated with a difference in the risk of rehospitalization. We found that the risk of readmission at 30 days was six-fold higher among those observed for less than 24 hours compared to those who were given at least a day of oral therapy prior to discharge (11). The only other independent predictor of readmission was a prior hospitalization for heart failure, suggesting that even those patients who are well-known to the team remain at exceedingly high risk of being rehospitalized with ADHF and therefore still warrant a trial of oral diuretics prior to discharge.
Although prolonged lengths of stay can certainly increase costs, they are still less costly than a hospital readmission. Furthermore, transitioning patients to oral diuretic therapy prior to the day of discharge doesn’t necessarily mean extending the length of stay if the team is proactive about making this transition once discharge within the next 24-48 hours become likely.
Bottom line: To reduce the risk of therapeutic failure, transition patients from IV to oral loop diuretics prior to the day of discharge in order to observe their response.
 |
Brent N. Reed, PharmD, BCCP, FAHADr. Reed is an associate professor in the Department of Pharmacy Practice and Science at the University of Maryland School of Pharmacy, and practices as a clinical pharmacy specialist in advanced heart failure at the University of Maryland Medical Center in Baltimore, MD. Follow him on his website or on Twitter @brentnreed. |
References
- Felker GM, Lee KL, Bull DA, et al. Diuretic strategies in patients with acute decompensated heart failure. N. Engl. J. Med. 2011;364:797–805.
- Aronson D, Burger AJ. The relationship between transient and persistent worsening renal function and mortality in patients with acute decompensated heart failure. J. Card. Fail. 2010;16:541–547.
- Teerlink JR, Cotter G, Davison BA, et al. Serelaxin, recombinant human relaxin-2, for treatment of acute heart failure (RELAX-AHF): a randomised, placebo-controlled trial. Lancet Lond. Engl. 2013;381:29–39.
- Packer M, O’Connor C, McMurray JJV, et al. Effect of Ularitide on Cardiovascular Mortality in Acute Heart Failure. N. Engl. J. Med. 2017;376:1956–1964.
- O’Connor CM, Starling RC, Hernandez AF, et al. Effect of nesiritide in patients with acute decompensated heart failure. N. Engl. J. Med. 2011;365:32–43.
- Schwartzenberg S, Redfield MM, From AM, Sorajja P, Nishimura RA, Borlaug BA. Effects of vasodilation in heart failure with preserved or reduced ejection fraction implications of distinct pathophysiologies on response to therapy. J. Am. Coll. Cardiol. 2012;59:442–451.
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2013;62:e147-239.
- Dikshit K, Vyden JK, Forrester JS, Chatterjee K, Prakash R, Swan HJ. Renal and extrarenal hemodynamic effects of furosemide in congestive heart failure after acute myocardial infarction. N. Engl. J. Med. 1973;288:1087–1090.
- Ellison DH, Felker GM. Diuretic Treatment in Heart Failure. N. Engl. J. Med. 2018;378:684–685.
- Heart Failure Society of America, Lindenfeld J, Albert NM, et al. HFSA 2010 Comprehensive Heart Failure Practice Guideline. J. Card. Fail. 2010;16:e1-194.
- Laliberte B, Reed BN, Devabhakthuni S, et al. Observation of Patients Transitioned to an Oral Loop Diuretic Before Discharge and Risk of Readmission for Acute Decompensated Heart Failure. J. Card. Fail. 2017;23:746–752.
Share this post:


Thanks for a really helpful article Brent! I often hear people choosing to give a furosemide infusion over bolus dosing because of an assumption it causes less of a drop in blood pressure. This is typically in a patient who has a systolic of around 100 where there is concern a drop in blood pressure may create a need for inotropic support. Is there any evidence to support this assumption? I have been unable to find any blood pressure data in the DOSE results.
That’s a great question, George, and an argument I’ve also heard for omitting boluses, even with the initiation of a continuous infusion. As you point out, changes in blood pressure or rates of symptomatic hypotension were not captured in the DOSE trial. One of the smaller studies to compare IV bolus and continuous infusion administration collected some data on blood pressure. In a study by Thomson, et al (PubMed ID 20206891, n=56), the rate of significant hypotension was no different between continuous infusions and IV boluses (35% vs. 37%, respectively, p=0.87) and there were also no differences in the number of antihypertensive doses held (7 vs. 8, p=0.98) or lowest systolic blood pressure values observed (98.9 vs. 93.8, p=0.14).
From a mechanistic standpoint, loop diuretics could cause hypotension via two mechanisms. The first would be due to functional venodilation, i.e., reductions in preload that could result in drops in stroke volume/cardiac output and thus mean arterial pressure. The risk of this is pretty low in HFrEF given the flatness of the Frank-Starling curve in those patients. The risk is probably a bit higher in HFpEF but I can’t say I’ve seen it in my own experience. The other mechanism would be an acute drop in intravascular volume due to rapid diuresis. The latter is certainly possible if one’s initial assessment is incorrect (i.e., less volume overloaded than expected) but otherwise, I feel like transcapillary refill is usually sufficient enough to prevent significant hypotension in most patients. There are probably select subgroups who may be more sensitive to rapid shifts in intravascular volume (again, HFpEF or those with delayed transcapillary refill, e.g., hypoalbuminemia).
When people express concern about administering a bolus, I’ll recommend either slowing the dose down (perhaps giving it over 15-20 minutes rather than an IV push) or if the plan is to initiate an infusion, giving a lower bolus dose than I would have normally recommended (since the main advantage of boluses in the latter scenario is to speed the time to clinical response).
Thanks for your question and for reading our blog!
Brent
Thanks for your reply Brent, that’s really helpful.